Paediatric patients with heterozygous familial hypercholesterolaemia treated with evolocumab for 80 weeks (HAUSER-OLE): a single-arm, multicentre, open-label extension of HAUSER-RCT - The Lancet Diabetes & Endocrinology
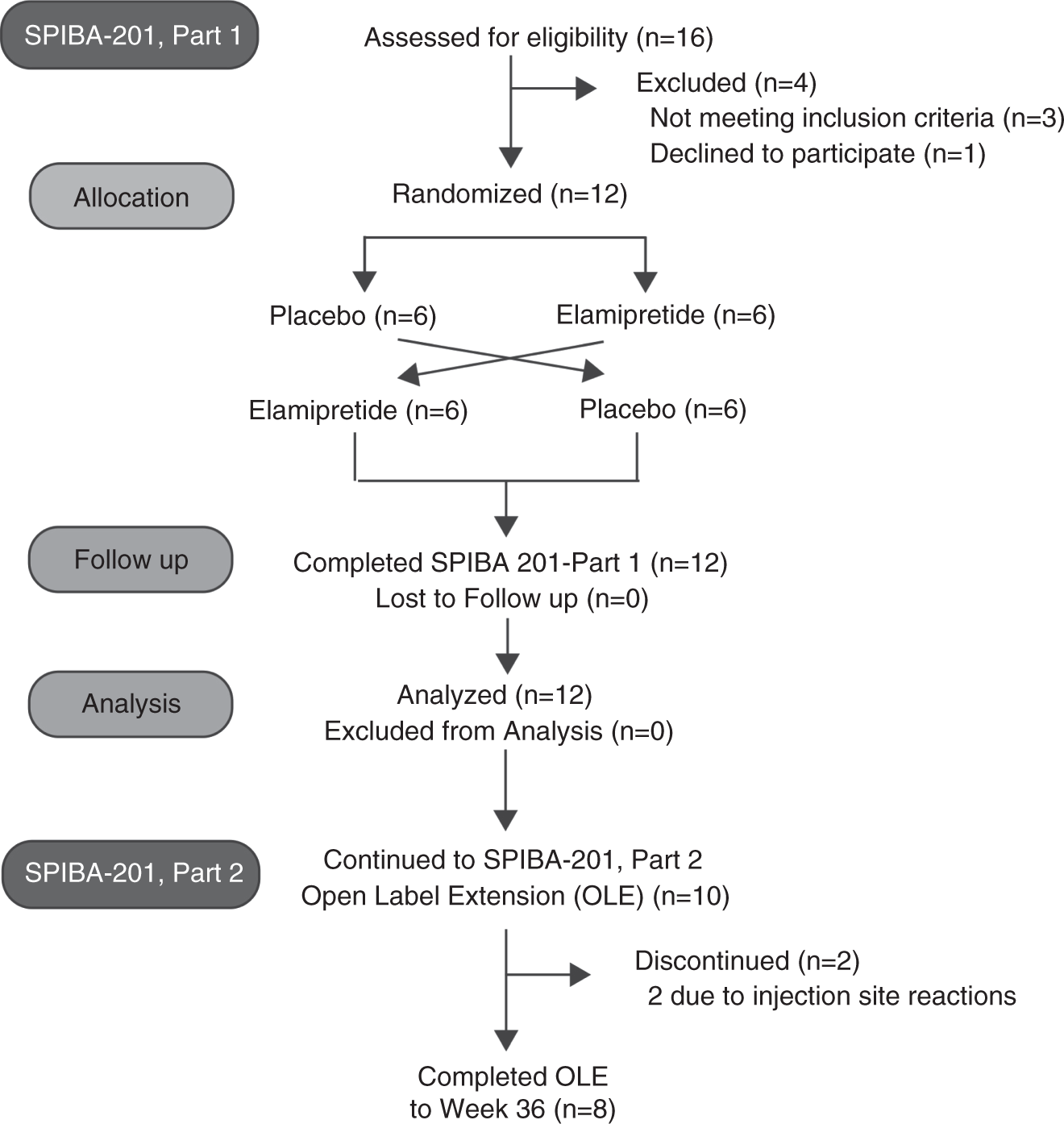
A phase 2/3 randomized clinical trial followed by an open-label extension to evaluate the effectiveness of elamipretide in Barth syndrome, a genetic disorder of mitochondrial cardiolipin metabolism | Genetics in Medicine