SOLVED: Define the following terms used in volumetric/ titrimetric analysis Standard solution b. Titrate/titrand/analyte C. Titrant d Endpoint Equivalence point f. Indicator g Titration h Titration error 2 What are the types

Warm-up n Define the term solution in your own words. n What are three examples of solutions? - ppt download

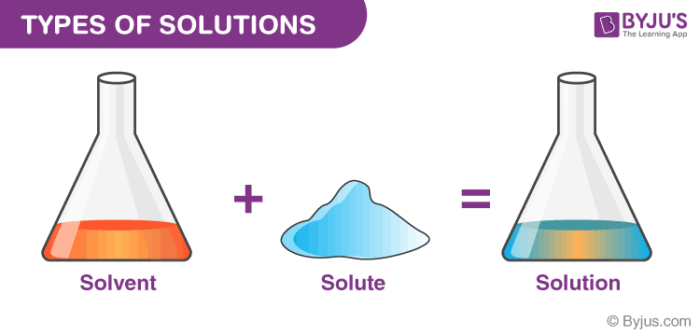
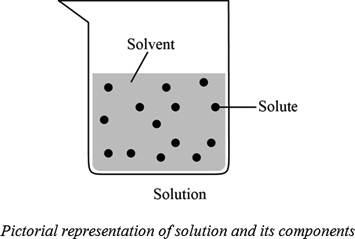
Define the following terms. 1.Solution-2. Solute- 3.solvent-4. Solubility-5. Unsaturated solution- 6. - Brainly.ph
Define the term solution. How many types of solutions are formed? Write briefly about each type with an example. - Sarthaks eConnect | Largest Online Education Community

2.3 Solutions. Objectives 9. Define solution, solute, solvent, and concentration 10. Explain the dissociation of water 11. Contrast properties of acids. - ppt download

Define the term 'molarity of a solution'. | 12 | SOLUTIONS | CHEMISTRY | DINESH PUBLICATION | Do... - YouTube

Chapter 15 Solutions Standard #6. I.What's a solution? A. Definition : A solution is a homogeneous mixture of two or more substances in a single physical. - ppt download

Q8 Define the term 1 Solute 2 Solvent 3 Solution with reference to addition of sodium chloride in wa...

Define the term solution. How many types of solutions are formed? Write briefly about each type with an example.
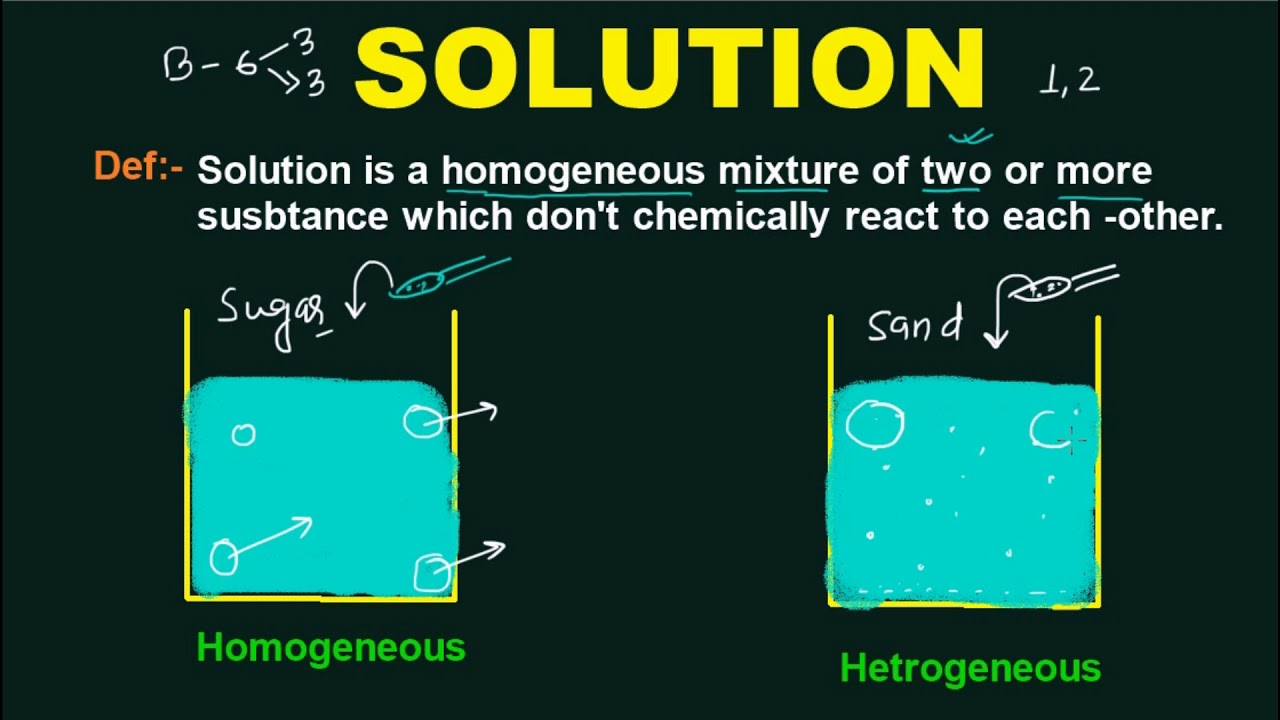
solution chemistry in hindi L-1| Definition + solution chemistry meaning,| | Eminent Guide - YouTube

Define the term solution. How many types of solutions are formed? Write briefly about each type with an example.