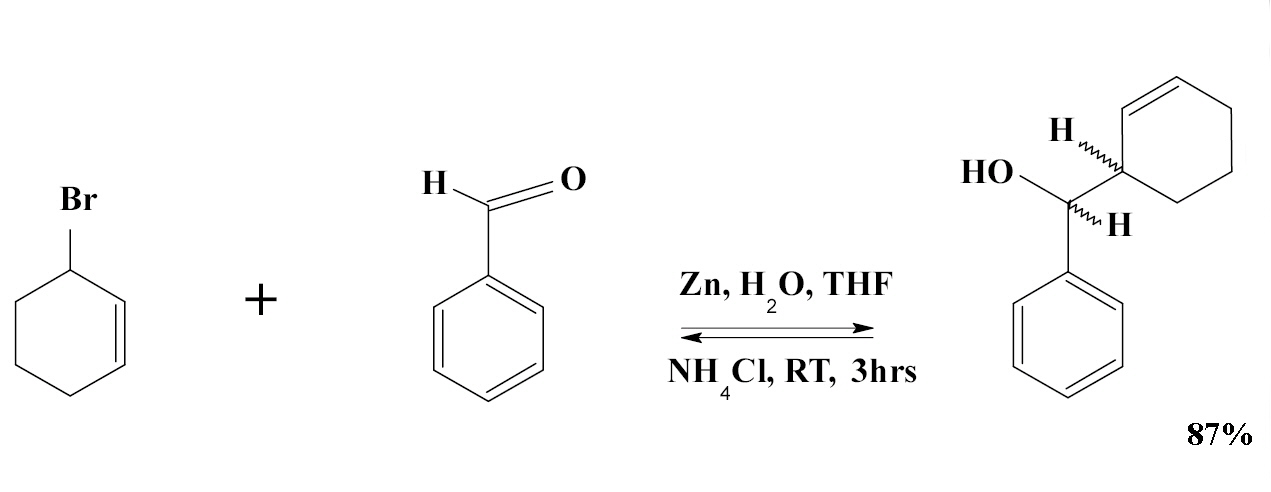
The Barbier−Grignard-Type Carbonyl Alkylation Using Unactivated Alkyl Halides in Water | Journal of the American Chemical Society

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society
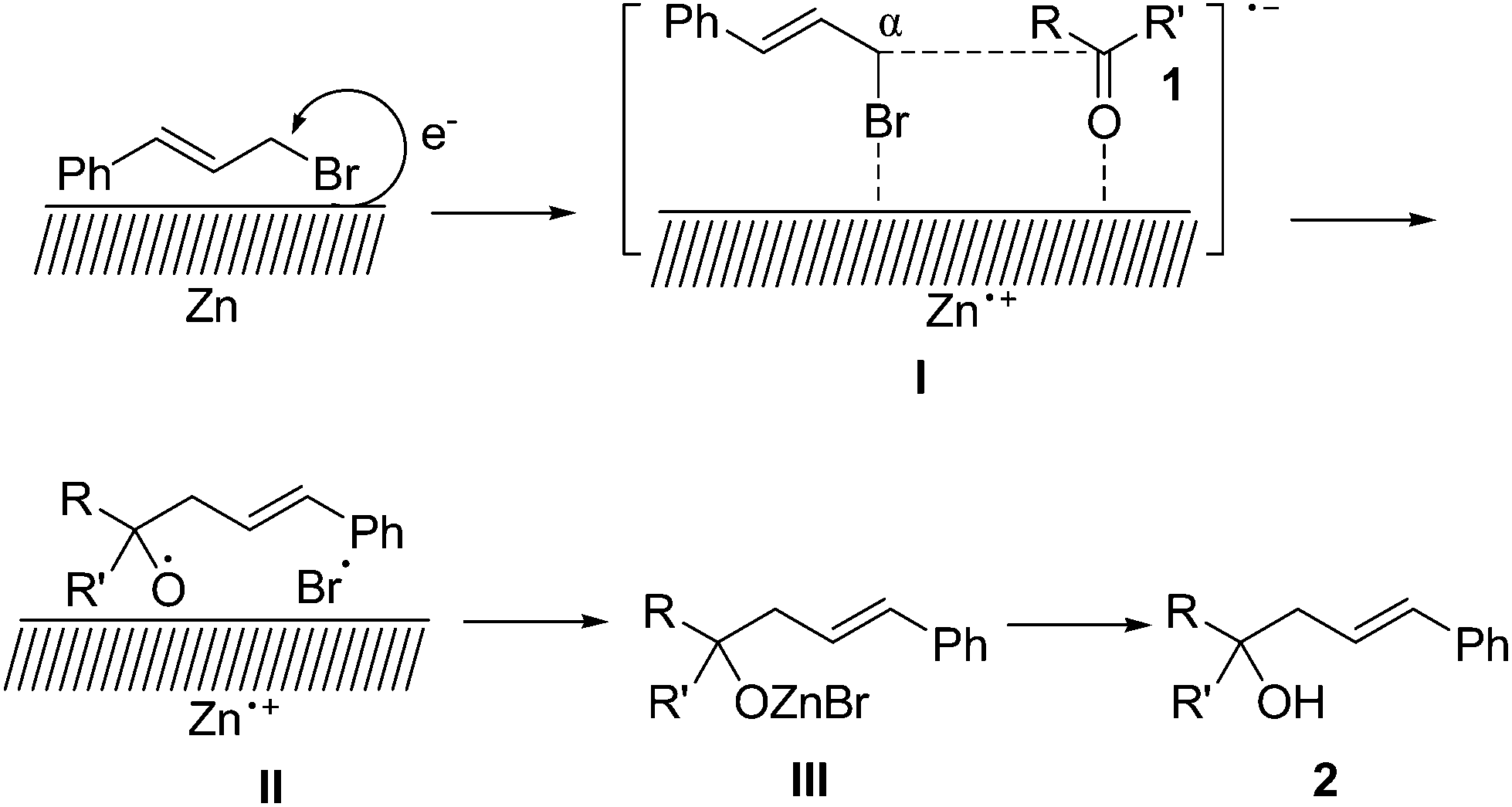
Zinc-mediated α-regioselective Barbier-type cinnamylation reactions of aldehydes, ketones and esters - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB00723J
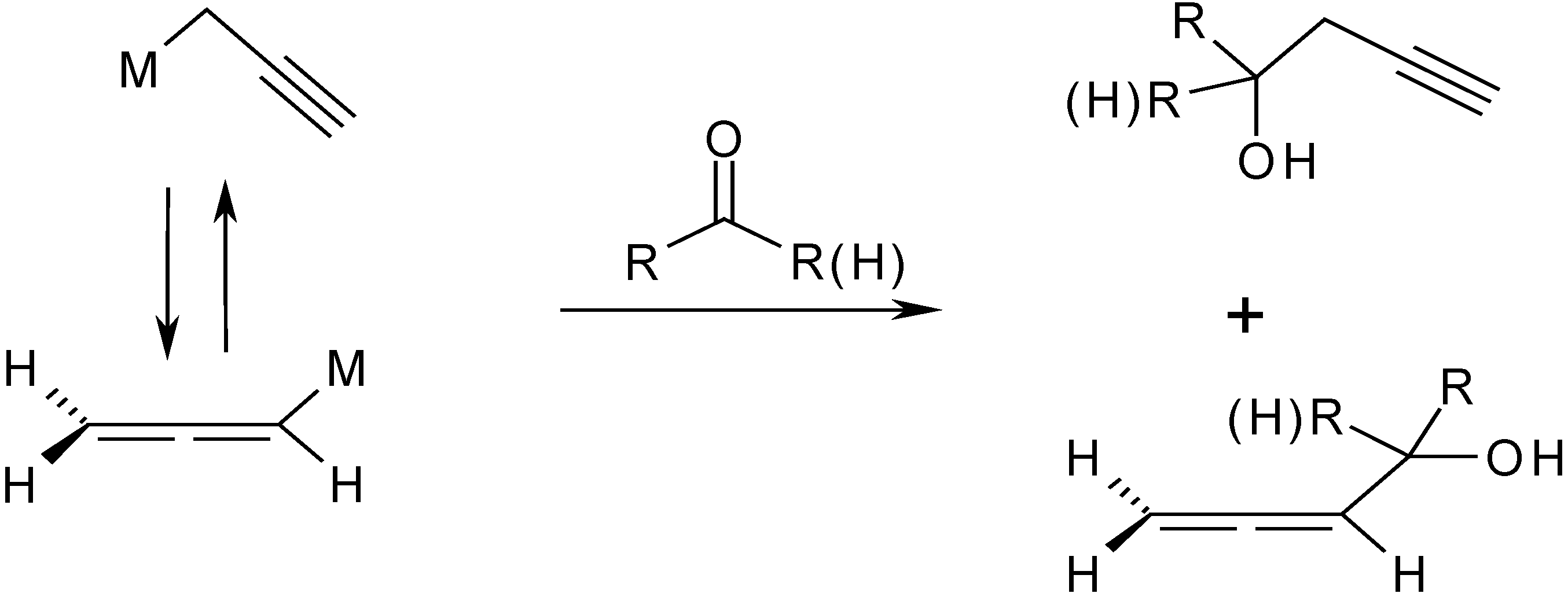
Molecules | Free Full-Text | Zn Mediated Regioselective Barbier Reaction of Propargylic Bromides in THF/aq. NH4Cl Solution

Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society
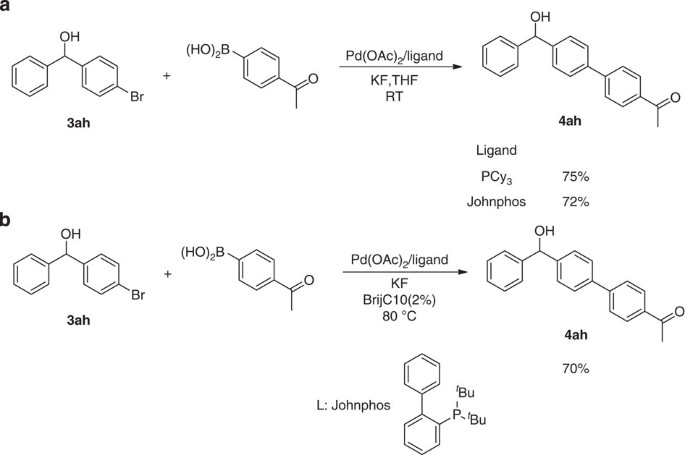
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

A Mechanochemical Zinc-Mediated Barbier-Type Allylation Reaction under Ball-Milling Conditions | The Journal of Organic Chemistry
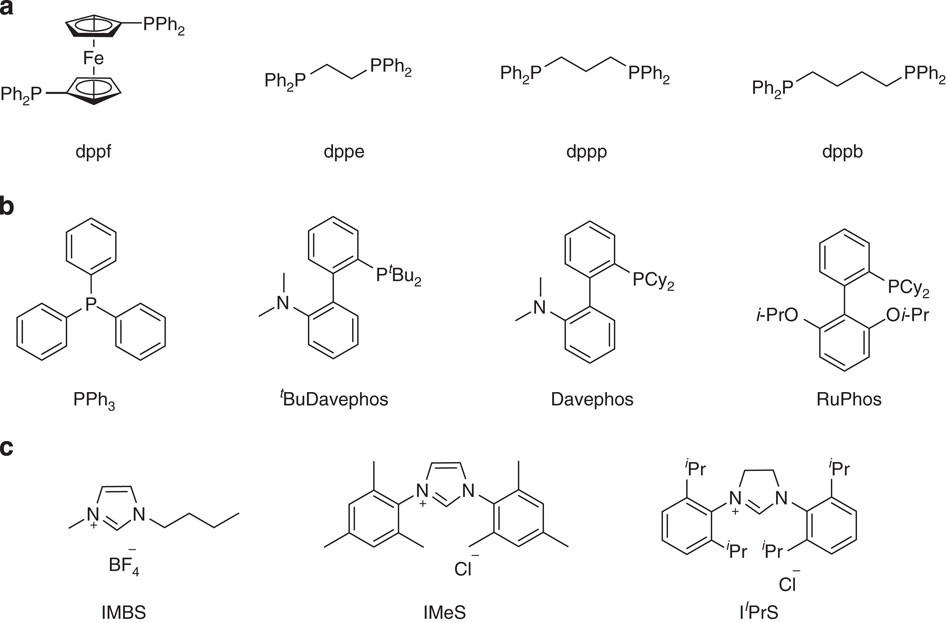
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
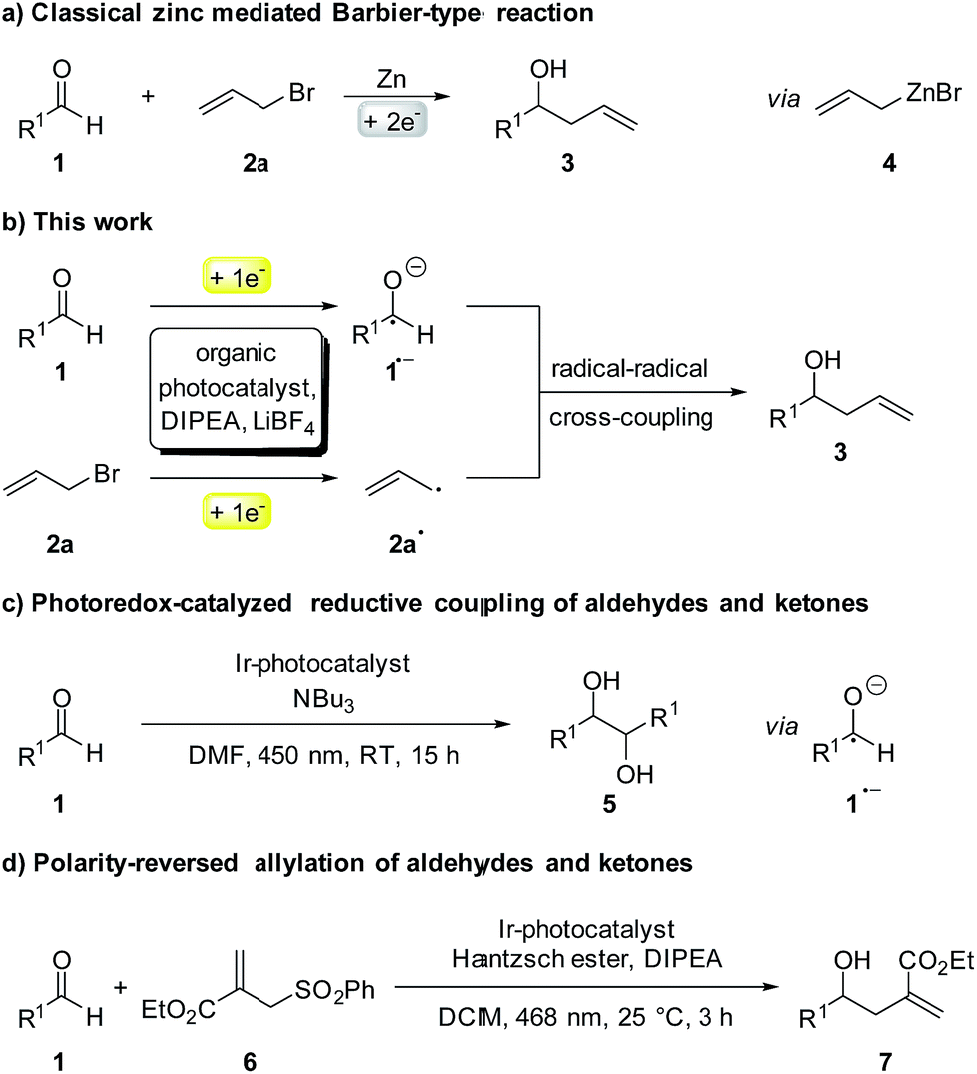
Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones - Chemical Science (RSC Publishing) DOI:10.1039/C8SC02038H